|
 Home:
Educational
Supplement: Section 1
Home:
Educational
Supplement: Section 1
Section 1
Breast
cancer clinical trials: Detecting modest but important advances
through increased statistical power
EDITOR'S
NOTE
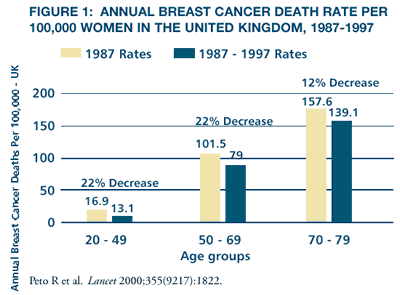
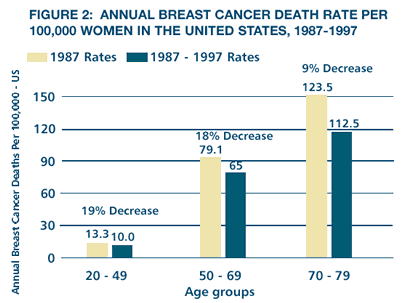
The
recently reported decline in breast cancer mortality in the United
States and United Kingdom (Figures 1 and 2) has been attributed
to multiple factors, particularly the increased use of screening
mammography, adjuvant chemotherapy and endocrine therapy with tamoxifen.
Sir Richard
Peto has estimated that in the United States alone, 10,000 fewer
women a year are dying from breast cancer because of these advances,
and he has predicted that over the next decade mortality rates will
continue to decline, as established improvements in adjuvant systemic
therapy are implemented in clinical practice.
Peto first proposed
the concept of large scale clinical trials to detect modest but
humanly important advances at the 1985 NIH Consensus Conference
on Early Breast Cancer. In an initial public presentation of what
was to become an ongoing series of key findings from an international
meta-analysis of randomized clinical trials, the first Breast Cancer
Overview revealed a sentinel groundbreaking finding about adjuvant
therapy that had a profound effect on the structure of breast cancer
clinical research.
Prior individual
randomized studies had demonstrated that adjuvant tamoxifen improved
disease-free survival, but no statistically significant impact on
mortality was evident. Many observers concluded that adjuvant systemic
therapy - particularly "cytostatic" endocrine therapy
- would only delay recurrence without affecting the long-term outcome.
Peto noted that there are two potential reasons that clinical trial
data fail to demonstrate an effect, such as mortality reduction:
1. The intervention
has no effect on the outcome, e.g., adjuvant tamoxifen does not
reduce breast cancer mortality.
or
2. The intervention
does have the effect, but the data are not statistically powered
to detect the effect, e.g., adjuvant tamoxifen does reduce mortality,
but there were not enough events - in this case, deaths - observed
in the individual trials to detect that effect.
The statistical
mantra for this concept is: "The lack of evidence of an effect
is not necessarily evidence of a lack of that effect. "Specifically,
researchers realized that one of the most salient features of research
planning was to focus on the expected number of measured events,
as opposed to just the size and follow-up of trials.
In what could
be viewed as a classic moment in breast cancer medicine, Peto's
1985 presentation demonstrated that combining the existing randomized
individual trial data on tamoxifen into a meta-analysis revealed
a clear-cut impact on mortality that was obscured by smaller trials
with relatively few deaths.
Increasing recognition
of the importance of adequate statistical power in clinical trials
has led to the evolution of large cooperative clinical research
groups with the infrastructure to conduct studies with a sufficient
number of events to detect moderate but important improvements in
outcome. In adjuvant trials, recurrences and deaths are usually
key measured events, and for example, it became clear that for lower-risk
study populations, more patients and longer follow-up would be required
to observe an adequate number of these events. A fascinating footnote
on this concept is the soon-to-be-reported ATAC adjuvant trial -
perhaps the largest individual randomized study ever conducted in
breast cancer - which has about ten times as many patients as the
largest trial in the original tamoxifen meta-analysis reported by
Peto in 1985.
(Figures 3A, 3B, 3C)

Figure
3A: Click here to enlarge
Page
1of 4
Next
page
Top
Additional
Sections:
1 | 2
| 3 | 4
| 5 | 6
| 7 | 8
| 9 | 10
| 11 | 12
|
|

