|

On September 19, 2005, after several decades of attending NSABP membership
meetings and listening in rapt attention as Bernie Fisher and his team
forced the field forward, I finally had the opportunity to sit at the dais with
clinical research leaders at the front of the room.
By way of background, in June, during a lunch break at our CME group’s
annual colorectal cancer Think Tank, I approached NSABP chairman Dr
Norman Wolmark with the idea of partnering on a special education symposium
covering the landmark adjuvant trastuzumab data that had just been
presented at the ASCO meeting in Orlando.
The idea was to invite key clinical investigators from the major cooperative
groups — BIG, NCCTG-Intergroup and BCIRG — that conducted the
adjuvant trastuzumab trials to join NSABP researchers in discussing where
we’ve been, where we are and where we’re heading in adjuvant therapy for
patients with HER2-positive tumors.
We proposed a special two-hour symposium during the NSABP group
meeting with the edited proceedings of that event published along with
individual interviews of the faculty members as an audio/print/web enduring
education piece for physicians. What made this idea even more enticing was
that the 2005 NSABP meeting was scheduled to take place in my hometown
of Baltimore (pronounced “Balamer” by natives).
I tossed this idea out to Norm without any idea how he would react, but after
munching thoughtfully a bit more on his salad, he said, “It seems like a short
turnaround time, but send me a proposal, and if you think this can be pulled
off, we’ll consider it.”
A couple weeks later, we were up and running with Charles Geyer as the
NSABP point person on the project. We were also fortunate enough to recruit
the “father” of trastuzumab, Dennis Slamon, along with Brian Leyland-Jones
and Peter Kaufman to join Norm, Chuck and Edward Romond from the
NSABP to serve as the faculty for this unique event. Two months later, as
we were wrapping up our planning for this meeting, my first homecoming
surprise occurred.
I was exchanging emails with John Mackey — a key figure in the BCIRG
— about an upcoming CME meeting, when I happened to ask him about the
current status of BCIRG trial 006, the fourth and perhaps most intriguing of
the international adjuvant trastuzumab trials.
Investigators had been telling me for months that this critical study was very
close to its first analysis and that the definitive presentation of the initial
data set was likely to occur at the San Antonio Breast Cancer Symposium in
December. Of course, the principal investigator for 006 is Dennis Slamon.
One of the reasons for the intense interest in 006 is that this BCIRG trial was
the only one of the four major studies that included a treatment arm without
an anthracycline — TCH (docetaxel, carboplatin, trastuzumab). Many
researchers, including Dr Slamon, were expecting this regimen to provide
equal or greater efficacy compared to AC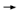 docetaxel/trastuzumab (AC docetaxel/trastuzumab (AC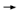 TH) but with little or no cardiac toxicity. TH) but with little or no cardiac toxicity.
To my surprise, Dr Mackey told me that, in fact, the requisite number of
events (recurrences) in 006 had just occurred, and the Independent Data
Monitoring Committee (IDMC) was to meet and review these data just a few
days prior to our NSABP event.
I digested this information and concluded that like the other adjuvant trastuzumab
studies, 006 was likely to show an important advantage to adding
the anti-HER2 antibody and that, as with other important trial results in
oncology over the last few years, some type of press release was likely to be
issued if the results were positive.
Sure enough, on September 15, just four days before the NSABP meeting, our
scientific staff retrieved a press release from the BCIRG website announcing
some intriguing findings — namely that patients on both the AC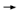 TH and
TCH arms of the study had experienced significantly fewer relapses than
patients on the AC TH and
TCH arms of the study had experienced significantly fewer relapses than
patients on the AC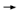 T arm. Interestingly, although the press release indicated
that there was not a statistically significant difference between the relapse rates
of the two trastuzumab arms, to the naked eye, the relative reduction with
TCH (39 percent) seemed less impressive than that of the AC T arm. Interestingly, although the press release indicated
that there was not a statistically significant difference between the relapse rates
of the two trastuzumab arms, to the naked eye, the relative reduction with
TCH (39 percent) seemed less impressive than that of the AC TH arm
(51 percent). TH arm
(51 percent).
Mama Mia! With Dr Slamon as part of our NSABP symposium, and hopefully
willing to discuss his perspective on these fresh data nuggets, we were in the
midst of a continuing medical education coup.
On this program, you will hear the results of that serendipitous timing as Dr
Slamon comments on how he interprets the findings released by the IDMC,
and this story will continue in December in San Antonio when the BCIRG
006 data will be presented by Dr Slamon as the initial plenary talk.
The second surprise came the day after the symposium, when I interviewed
Dr Norman Wolmark. (Interviews with Dr Slamon and Dr Leyland-Jones are
also on this program, and interviews with Drs Geyer and Kaufman will be included on our next issue of Breast Cancer Update. Dr Romond was interviewed
this summer for our series.)

Dr Wolmark has been a regular interviewee for our series since 1991, and
working with this research giant over the years, I have found him — like Dr
Fisher — not only to have encyclopedic oncologic knowledge but also to be a
champion of patients and the clinical trials process.
In the process of setting up the NSABP education symposium, I had somewhat
timidly sent Dr Wolmark an email commenting on our CME group’s experience
with education programs on lung cancer, a disease that takes 155,000
lives a year in this country alone. The few adjuvant trials in lung cancer that
have been conducted are woefully underpowered, and it was only in the last
three years that four “large” randomized studies had finally confirmed that
adjuvant chemotherapy has a very significant impact on relapse rate and overall
survival. The clinical benefit of this treatment strategy, in fact, is similar in
magnitude to or greater than that has been observed in breast cancer trials
since the 1979 NIH consensus conference.
My email to Dr Wolmark noted that these four critical lung cancer trials were
conducted largely outside the United States and comprised an aggregate total of
about 3,500 patients. With great humility, I asked Dr W if the NSABP would
ever consider becoming involved in adjuvant lung cancer trials and perhaps
developing working relationships with thoracic surgeons in the same manner
that they have so successfully accomplished with breast and colorectal surgeons.
At the completion of his interview in Baltimore, I again raised my lung cancer
question — initially off the record — and this led to a very interesting interlude
of comments. Yes, the NSABP would potentially be willing to consider
lung cancer trials if there was an imprimatur that this would be in the national
interest. However, my query opened up a breached levee of criticism by Dr W
directed at the NCI and its director Andrew von Eschenbach.
This brief thunderstorm of emotion left me wide eyed and slack jawed, and
I suggested to Norm that if he really felt so strongly about this, maybe we
should include his commentary on our audio program, editing out some of the
colorful language, of course. Norm paused momentarily and, to my great satisfaction,
agreed to allow this blistering commentary on the enclosed program.
That evening, dining with my family at Bo Brooks Crab House, happily
cracking away at Balamer’s finest bay-seasoned crustaceans, I reflected on my
homecoming visit and felt a deep sense of satisfaction and gratitude to have
had the honor to work so closely with the NSABP leadership and membership
on this exciting project, but even more importantly, it was truly heartening to
know that our CME group had the opportunity to assist clinicians in practice
obtain the most up-to-date information on a fascinating, groundbreaking and
definitely here-to-stay targeted biologic agent.
— Neil Love, MD
NLove@ResearchToPractice.net
| The PowerPoint slides presented at the NSABP trastuzumab education
symposium are included on the enclosed third audio CD and are posted at www.BreastCancerUpdate.com/NSABP. |



|

