
 |
||||||||
COMMENTS FROM BREAST CANCER INVESTIGATORS
![]() NSABP-B-35 and IBIS-II are important trials,
both comparing anastrozole and tamoxifen in
postmenopausal patients with DCIS. Aromatase
inhibitors have already proved to have a significant
effect in invasive cancer, and it’s highly
likely they will affect DCIS as well.
NSABP-B-35 and IBIS-II are important trials,
both comparing anastrozole and tamoxifen in
postmenopausal patients with DCIS. Aromatase
inhibitors have already proved to have a significant
effect in invasive cancer, and it’s highly
likely they will affect DCIS as well.
We know that the majority of DCIS lesions are likely to be ER-positive. Craig Allred has shown that age-per-age, tumor-for-tumor, DCIS is even more likely to be ER-positive than invasive cancer. If that’s true, then we have even more reason to be optimistic about the studies of aromatase inhibitors in DCIS.
We have viewed tamoxifen as a highly appropriate option for treating a patient with ER-positive DCIS since the NSABP-B-24 trial.
However, when we consider risks, benefits and quality-of-life issues, it’s common for our New York patients to demur, so we probably have one of the lowest percentages of patients with ER-positive DCIS on tamoxifen in the country.
The same can be seen in our prevention setting, in which we’ve not been successful in getting patients to take tamoxifen.
The two most obvious concerns about tamoxifen in these settings are endometrial cancer and gynecological events. Even when we provide the raw numbers on how infrequent those events are, because we are talking about minimal, if any, impact on long-term survivorship and moderate impact on local control, it simply is not an attractive option.
We’d like more information about DCIS and aromatase inhibitors, but since the initial publication of the ATAC data, aromatase inhibitors have become our endocrine therapy of choice for postmenopausal patients with ER-positive, invasive cancers. That literally happened overnight, like gangbusters, and so a “bleed over” to postmenopausal patients with DCIS is natural.
In my clinical practice, it’s clear that the aromatase inhibitors are vastly better tolerated than tamoxifen in postmenopausal patients. Our surgeons are beginning to give first-line endocrine therapy without a mandatory consult from medical oncology. We perform bone density tests before we start our patients on aromatase inhibitors, and treating these patients has been satisfying.
— Patrick I Borgen, MD
![]() There is still uncertainty about what constitutes
adequate local treatment, and much remains to
be done to delineate the various subtypes of DCIS
and to ascertain the minimum adequate treatment
for each. However, the case for hormonal
treatment of oestrogen or progesterone receptor-positive
DCIS is already strong and, accordingly,
we have focused our attention on which hormonal
treatment — tamoxifen or anastrozole — is best
for this subgroup of patients. For IBIS II, women
with DCIS have up to 6 months to decide about
entry, so there is plenty of time for discussion
and no rush to make this important decision.
There is still uncertainty about what constitutes
adequate local treatment, and much remains to
be done to delineate the various subtypes of DCIS
and to ascertain the minimum adequate treatment
for each. However, the case for hormonal
treatment of oestrogen or progesterone receptor-positive
DCIS is already strong and, accordingly,
we have focused our attention on which hormonal
treatment — tamoxifen or anastrozole — is best
for this subgroup of patients. For IBIS II, women
with DCIS have up to 6 months to decide about
entry, so there is plenty of time for discussion
and no rush to make this important decision.
— Jack Cuzick et al.
Lancet 2003;362:832-3.
![]() The IBIS-II prevention trial compares the
aromatase inhibitor anastrozole to placebo. We’ll
all be surprised if anastrozole does not reduce
the incidence of breast cancer. Although we’re
currently fixated on the bone effects associated
with the aromatase inhibitors, I believe we will
find that with the new, powerful bisphosphonates,
the bone effects will not be a long-term
problem.
The IBIS-II prevention trial compares the
aromatase inhibitor anastrozole to placebo. We’ll
all be surprised if anastrozole does not reduce
the incidence of breast cancer. Although we’re
currently fixated on the bone effects associated
with the aromatase inhibitors, I believe we will
find that with the new, powerful bisphosphonates,
the bone effects will not be a long-term
problem.
One of the strengths of the IBIS-II prevention trial is that it will help identify women for whom we need to do bone scans and those with whom we need to use bisphosphonates. In the separate bone subprotocol of the IBIS-II prevention trial, women with a high baseline bone mineral density (BMD) won’t receive a bisphosphonate, women with a low baseline BMD will automatically receive a bisphosphonate and women with a baseline BMD in the midrange will be randomly assigned to a bisphosphonate or placebo.
— J Michael Dixon, MD
SUPPORTING PROTOCOL INFORMATION
Background Information
DCIS was once a rare diagnosis, but it has become increasingly common following the advent of mammographic screening. Radiotherapy has been shown to reduce local recurrence rates by 60-70%, but there is still uncertainty as to which group of patients require this rather intensive treatment. Adjuvant endocrine therapy has also been advocated, and there are two trials which have addressed this issue. The North American NSABP-B-24 trial looked at the value of giving tamoxifen to women who had received radiotherapy. A highly significant 37% reduction in recurrence rate was found. However, the recently completed UK trials have not been so positive, reporting a non-significant 17% reduction in recurrence. The clear benefit of tamoxifen in the adjuvant studies, even for early stage 1 disease also provides strong support for an effect of hormonal therapy in DCIS. The focus of the current trial will be to evaluate whether the aromatase inhibitor anastrozole has advantages over tamoxifen, either in terms of reduced recurrence rates or lower side effects.
Correlative Science Program
A set of representative diagnostic H&E stained slides plus the original hormone receptor assay slides (ER and/or PgR) will be required from all patients for central review. Paraffin blocks containing representative areas of the tumour will also be requested.
Diagnostic slides and paraffin blocks will be requested for all breast, endometrial or ovarian cancers developing after trial entry. These samples will be used for central pathology review and marker studies, and will remain the property of the Steering Group, who will be responsible for deciding how they will be used in any further projects.

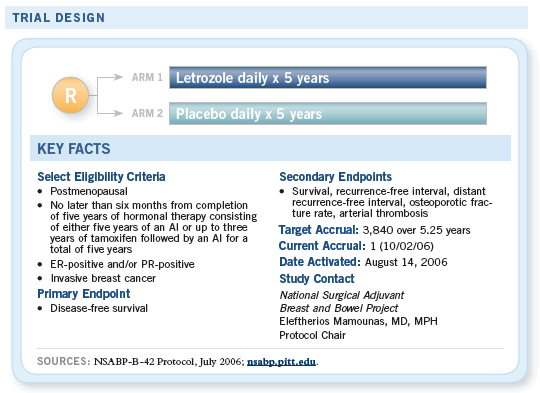
COMMENTS FROM BREAST CANCER INVESTIGATORS
![]() NSABP-B-42 just opened. It has a sample
size of about 3,800, and of course one of
the questions that remains unanswered is the
duration of an aromatase inhibitor.
NSABP-B-42 just opened. It has a sample
size of about 3,800, and of course one of
the questions that remains unanswered is the
duration of an aromatase inhibitor.
We went through this process and it took us years to determine the optimum duration of tamoxifen therapy, and at the end of the day there was enormous surprise from the B-14 data that not only is 10 years not as good as five, but it is also somewhat detrimental.
We believe it’s important to address the duration of an aromatase inhibitor, and this is what NSABP protocol B-42 will be doing.
The data with aromatase inhibitors from the multiple trials have all been positive. The duration question remains relatively unaddressed. We have seen trials that have introduced aromatase inhibitors after a period of tamoxifen and have shown an advantage.
We’ve seen direct head-on comparisons between aromatase inhibitors and tamoxifen up front also showing an advantage, and we’re waiting to see the results of a trial that starts with an aromatase inhibitor and sequences it with tamoxifen.
— Norman Wolmark, MD
![]() The problem with the extended letrozole trial
(NCIC-CTG-MA17) was that the patients were
unblinded at 2.4 years, and because most
patients then switched over to the active agent,
we will never know with any certainty what would
have happened had they been unblinded at five
years.
The problem with the extended letrozole trial
(NCIC-CTG-MA17) was that the patients were
unblinded at 2.4 years, and because most
patients then switched over to the active agent,
we will never know with any certainty what would
have happened had they been unblinded at five
years.
That is a shame because we are going to be treating these patients for five years, so it would have been nice to know the differences in toxicity and efficacy between the two arms. The data for one or two years are complete because most of the patients had gone through those years. There were a lot of data in year three, a modest amount in year four and almost no data for the fifth year.
An analysis of relapse risk within each year could then be performed. This was possible not only for years one and two but also for year three, when it seems that the relative benefit was greater, which is interesting and reassuring. That was also the case in year four. That analysis used year-by-year hazards to determine whether benefit was attenuating, staying as strong or becoming stronger.
Although we will never know what it would have been if the trial had been unblinded at five years, we are somewhat reassured by the results of this analysis that going beyond 2.4 years of treatment is reasonable.
— Peter M Ravdin, MD, PhD
SUPPORTING PROTOCOL INFORMATION
Background Information
In the adjuvant setting, AIs have demonstrated activity in three distinct clinical situations. In the first situation, an AI was compared to tamoxifen as initial adjuvant hormonal therapy in patients with resected operable breast cancer.
The ATAC trial demonstrated that 5 years of anastrozole significantly improved disease-free survival (DFS) when compared to 5 years of tamoxifen. More recently, the BIG 1-98 trial also demonstrated improved DFS as well as distant DFS for 5 years of letrozole compared to 5 years of tamoxifen.
In the second situation, an AI was compared to tamoxifen in patients who had already received 2- 3 years of adjuvant tamoxifen. In three randomized trials (the IES trial [International Exemestane Study], the ABCSG-8/ARNO 95 trial, and the ITA trial [Italian Tamoxifen vs Anastrozole]), 2-3 years of an AI (exemestane or anastrozole) improved disease-free survival compared to 2-3 years of tamoxifen in patients who had already completed 2-3 years of tamoxifen therapy.
In the third clinical situation, an AI was evaluated as extended adjuvant hormonal therapy following completion of 5 years of adjuvant tamoxifen. The NCIC-MA17 trial compared 5 years of letrozole with 5 years of placebo in patients who had already completed 5 years of adjuvant tamoxifen and demonstrated significant improvement in disease-free survival in favor of the group that received the AI...
Based on the results from these trials, AIs are increasingly utilized as adjuvant therapy in these three clinical situations. At this time, there are no available results from trials that directly compare these different approaches for using AIs. Thus, the best setting for the adjuvant use of AIs cannot be readily determined at present...
As the adjuvant use of AIs continues to expand, the question of optimal duration needs to be definitively addressed. Whether less than 5 years of an AI given as up-front adjuvant therapy is as effective as 5 years is a question that is unlikely to be addressed, given that in the pivotal ATAC trial, 5 years of anastrozole were found to be superior to 5 years of tamoxifen. On the other hand, whether prolonged administration of an AI beyond 5 years will result in additional benefit is an important and clinically relevant question that is currently not being addressed in any clinical trial. It is quite possible that resistance to AIs in this setting may develop at different time intervals than resistance to tamoxifen...
Similarly, in the clinical situation where the AI is given for 2-3 years following 2-3 years of adjuvant tamoxifen, it is also unknown whether continuing the AI for more than 2-3 years might result in additional benefit...
Finally, no data exist on the optimal duration of AIs when used as extended adjuvant therapy after 5 years of tamoxifen. Five years of therapy was arbitrarily selected in the MA17 trial, but whether shorter therapy could be as effective or whether longer therapy could be more effective are questions that need to be addressed.
Correlative Science Program
We will be banking paraffin blocks from the primary tumor (or positive lymph node) in order to have an opportunity to evaluate promising markers in the future, if they become available. At this point, there are some data that suggest a differential benefit from an AI over tamoxifen based on the molecular profile of tumor cells. These data suggest that a certain subset of breast cancer patients might not derive maximum benefit from five years of tamoxifen and derive greater benefit from an AI.
It could be hypothesized that such patients may derive greater benefit if the AI is given for a longer duration. We will examine ER, PR, and HER2 by standardized central assays. We will explore quantitation of ER and PR by an image analysis program to examine the correlation between expression levels and benefit from the longer duration of an AI.
Abnormalities involving cofactors for ER signaling such as AIB1 may result in resistance to tamoxifen but not to an AI. Patients with such tumors can be hypothesized to benefit even more with a longer duration of an AI.


COMMENTS FROM BREAST CANCER INVESTIGATORS
![]() As the safety data for the aromatase inhibitors
are emerging, we see they are quite different. In
the package insert for exemestane, a small but
definite increased risk of cardiac dysfunction is
noted. If you consider the letrozole data from
the BIG trial, at 25 months a small but definite
increased risk of cerebrovascular accident and
myocardial infarct is evident.
As the safety data for the aromatase inhibitors
are emerging, we see they are quite different. In
the package insert for exemestane, a small but
definite increased risk of cardiac dysfunction is
noted. If you consider the letrozole data from
the BIG trial, at 25 months a small but definite
increased risk of cerebrovascular accident and
myocardial infarct is evident.
However, in the 68-month follow-up data for the ATAC trial, we see none of those risks with anastrozole. If you examine the cardiac deaths, it is 49 versus 46, and cerebrovascular accidents are substantially reduced with anastrozole compared to tamoxifen.
An interesting study presented at the 2005 San Antonio Breast Cancer Symposium evaluated 90 healthy, postmenopausal volunteers who received, in a blinded fashion, up to 24 weeks of either anastrozole, letrozole or exemestane. When the effects on the lipids were examined, they were found to be totally different.
We have to be aware of the different effects and realize that not all aromatase inhibitors are alike and that it does matter which one we select.
— Aman Buzdar, MD
![]() A significantly reduced risk of thromboembolic
disease was observed for all three AIs compared
with tamoxifen. Anastrozole is, at this point, the
only AI with a detailed benefit-risk profile from
over 5 years’ follow-up in the adjuvant setting.
A significantly reduced risk of thromboembolic
disease was observed for all three AIs compared
with tamoxifen. Anastrozole is, at this point, the
only AI with a detailed benefit-risk profile from
over 5 years’ follow-up in the adjuvant setting.
Thus far, no apparent CV-safety concerns have emerged. Preliminary data on letrozole and exemestane suggest that longer follow-up is needed for these two AIs before being able to fully assess their respective long-term CV toxicity profile.
The present differences in CV-safety profiles suggest that third-generation AIs should not be considered as equivalents in clinical practice.
— Jean Marc Nabholtz, Joseph Gligorov.
Drug Saf 2006;29(9):785-801.
![]() There may be important clinical differences
between the AIs. However, data from direct
comparative clinical trials are limited, and making
comparisons across trials is difficult given differences
in design, methodology, patients, and
endpoints. At the present time, the choice of
an AI for clinical use should be based on the
strength of the data within the distinct clinical
scenarios: neoadjuvant therapy, adjuvant therapy,
or advanced/metastatic disease.
There may be important clinical differences
between the AIs. However, data from direct
comparative clinical trials are limited, and making
comparisons across trials is difficult given differences
in design, methodology, patients, and
endpoints. At the present time, the choice of
an AI for clinical use should be based on the
strength of the data within the distinct clinical
scenarios: neoadjuvant therapy, adjuvant therapy,
or advanced/metastatic disease.
— John W Berry, MD.
Clin Ther 2005;27(11):1671-84.
![]() The aromatase inhibitor I would choose for
initial treatment would be either anastrozole
or letrozole. Following two to three years of
tamoxifen, it would be exemestane or anastrozole,
and after five years of tamoxifen, it would
be letrozole.
The aromatase inhibitor I would choose for
initial treatment would be either anastrozole
or letrozole. Following two to three years of
tamoxifen, it would be exemestane or anastrozole,
and after five years of tamoxifen, it would
be letrozole.
My expectation is that the aromatase inhibitors, as a class, are going to be active in all of those settings, but we don’t know that for sure and subtle differences in the adjuvant setting sometimes emerge in surprising ways. So at this time, I would favor using one of the aromatase inhibitors that has been used in clinical trials.
— Robert W Carlson, MD
![]() We don’t know what the appropriate approach
is to selecting one of the three aromatase inhibitors
in the up-front setting. I have the good
fortune of chairing a key study to this regard.
We don’t know what the appropriate approach
is to selecting one of the three aromatase inhibitors
in the up-front setting. I have the good
fortune of chairing a key study to this regard.
The MA27 study will complete accrual in 2006, and it is addressing precisely that question of whether there is an optimal aromatase inhibitor. The randomization is between the steroidal exemestane and the nonsteroidal anastrozole.
In the meantime, there are ample data to say these compounds are different in terms of their biochemical and preclinical effects. But in the clinic, with the present data, there is no evidence of a wide difference between these drugs. So I think that one has to restrict one’s choices to the approved therapies by the regulatory agencies and the published evidence-based data.
— Paul E Goss, MD, PhD
![]() As time goes on, there is less and less of a
distinction to be made between the aromatase
inhibitors. Up front, I don’t have a strong preference.
There are certainly data for anastrozole
and letrozole.
As time goes on, there is less and less of a
distinction to be made between the aromatase
inhibitors. Up front, I don’t have a strong preference.
There are certainly data for anastrozole
and letrozole.
I tend to use anastrozole simply because it has longer safety data. There we have the largest number of patients that have been followed, so in my mind, there’s more confidence in the safety profile.
— Debu Tripathy, MD

SOFT KEY FACTS
Select Eligibility Criteria
Stratification
Other Therapy
Primary Endpoint
Secondary Endpoints
Target Accrual: 3,000
Current Accrual: 767 (09/30/06)
Date Activated: August 4, 2003
Study Contacts
International Breast Cancer Study Group
Prudence Francis, MD, Protocol Chair
Breast International Group
Prudence Francis, MD, Protocol Chair
Cancer and Leukemia Group B
Gini Fleming, MD, Protocol Chair
TEXT KEY FACTS
Select Eligibility Criteria
Stratification
Other Therapy
Primary Endpoint
Secondary Endpoints
Target Accrual: 1,845
Current Accrual: 1,148 (09/30/06)
Date Activated: August 4, 2003
Study Contacts
International Breast Cancer Study Group
Olivia Pagani, MD, Protocol Chair
Breast International Group
Olivia Pagani, MD, Protocol Chair
Prudence Francis, MD, Protocol Chair
PERCHE KEY FACTS
Select Eligibility Criteria
Stratification
Primary Endpoint
Secondary Endpoints
Target Accrual: 1,750 within seven years
Current Accrual: Not reported
Date Activated: August 4, 2003
Study Contacts
International Breast Cancer Study Group
Rosalba Torrisi, MD, Protocol Chair
Breast International Group
Rosalba Torrisi, MD, Protocol Chair
COMMENTS FROM BREAST CANCER INVESTIGATORS
![]() For premenopausal women with endocrine-responsive
disease, we initiated three adjuvant
trials in August 2003: the Suppression of
Ovarian Function Trial (SOFT), the Tamoxifen
and Exemestane Trial (TEXT) and the Premenopausal
Endocrine Responsive Chemotherapy Trial
(PERCHE).
For premenopausal women with endocrine-responsive
disease, we initiated three adjuvant
trials in August 2003: the Suppression of
Ovarian Function Trial (SOFT), the Tamoxifen
and Exemestane Trial (TEXT) and the Premenopausal
Endocrine Responsive Chemotherapy Trial
(PERCHE).
SOFT will compare tamoxifen alone to ovarian function suppression with tamoxifen and ovarian function suppression with exemestane. This trial was designed specifically for oncologists who view tamoxifen as standard therapy.
TEXT will compare ovarian function suppression with tamoxifen to ovarian function suppression with exemestane. These patients may or may not receive chemotherapy.
PERCHE will determine whether adjuvant chemotherapy is necessary. Premenopausal women are randomly assigned to chemotherapy or no chemotherapy. Adjuvant chemotherapy selection is left entirely up to the investigator, and endocrine therapy consists of ovarian function suppression with tamoxifen or exemestane. Patients may also be randomly assigned to TEXT for endocrine therapy.
— Aron Goldhirsch, MD
![]() TEXT assumes that women need or should
undergo ovarian suppression as part of their
treatment protocol. The randomization is then
between tamoxifen and an aromatase inhibitor.
We tend to think of tamoxifen as the standard
of care for young, premenopausal women with
breast cancer, and the big controversy is whether
or not ovarian suppression or ovarian ablation
has a role.
TEXT assumes that women need or should
undergo ovarian suppression as part of their
treatment protocol. The randomization is then
between tamoxifen and an aromatase inhibitor.
We tend to think of tamoxifen as the standard
of care for young, premenopausal women with
breast cancer, and the big controversy is whether
or not ovarian suppression or ovarian ablation
has a role.
In fact, a fair amount of information exists to suggest that it does have a role. First, the Oxford overview with oophorectomy/ovarian irradiation suggests that, by itself, this is very good adjuvant therapy. If you go back to those old trials, you see approximately a 10 percent absolute change in mortality. Second, we have a lot of information comparing ovarian suppression, with or without tamoxifen, to chemotherapeutic regimens in patients with receptor-positive tumors. In many European trials, those two approaches are basically equivalent, although they are imperfectly conducted trials. Third, we have trials that have examined whether or not you should use ovarian suppression after chemotherapy. The results are mixed right now. The trials in aggregate don’t show a clear-cut advantage for this approach. However, from the point of view of retrospective subset analyses, a feeling does emerge from a couple of the trials that perhaps the younger women who were premenopausal at the end of chemotherapy and then received ovarian suppression are the ones who might be helped the most.
The problem with all of these trials is that they started in the late 1980s before we recognized that tamoxifen was a valid choice for young women with breast cancer. All of these studies lacked five years of tamoxifen, which is now the standard of care. I believe that is a fundamental flaw of these trials that we wish had been addressed.
Putting this all together is very complicated, and I don’t think I know the answer to the question. That is one reason why I’m such a strong proponent of SOFT, which evaluates tamoxifen versus ovarian suppression or ablation with tamoxifen versus ovarian suppression or ablation with an aromatase inhibitor. I believe this trial would help us to answer some of these questions that are a terrific struggle for us.
— Nancy E Davidson, MD
![]() The IBCSG is coordinating a series of three
nested trials: SOFT, PERCHE and TEXT. These
trials address what is probably the most important
conceptual question in premenopausal
breast cancer right now: Beyond tamoxifen, does
planned ovarian suppression benefit patients?
The IBCSG is coordinating a series of three
nested trials: SOFT, PERCHE and TEXT. These
trials address what is probably the most important
conceptual question in premenopausal
breast cancer right now: Beyond tamoxifen, does
planned ovarian suppression benefit patients?
In particular, does it benefit women who receive chemotherapy or who don’t receive chemotherapy, and if a woman experiences chemotherapy-related amenorrhea, does she still need ovarian suppression? These are important trials that offer a wonderful opportunity for community oncologists to participate in answering this critical question.
Currently, I consider ovarian suppression for two groups of patients. The first group includes patients at high risk — multiple positive nodes, very high-risk tumors — and particularly young women, less than 35 or 40 years of age, who may not go into menopause with chemotherapy. The other group includes women who are at the opposite end of the spectrum — very low-risk tumors, smaller tumors, node-negative — for whom the benefits of chemotherapy are small. With these women, I present ovarian suppression as an option, not necessarily in addition to chemotherapy but perhaps even instead of it.
— Harold J Burstein, MD, PhD
Editor’s Note:
Let’s get this thing done.
Prologue:
The adjuvant trastuzumab clinical research to
practice model
- Select publications
Breast Cancer Clinical Trials:
Neoadjuvant Therapy