| You
are here: Home: BCU 6|2001: Section 2
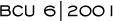
OVARIAN SUPPRESSION PLUS AROMATASE INHIBITORS
We need to study aromatase inhibitors in premenopausal women, because
there is almost no data. We used to think tamoxifen wouldn’t
work in premenopausal women, but it does. However, it doesn’t
work as well as in an estrogen-deficient environment. We have learned
over the years that suppressing ovarian function with tamoxifen
in a premenopausal patient with metastatic breast cancer gives maximum
benefit. We need studies with the AIs plus or minus ovarian suppression.
There have been some small studies of ovarian suppression, making
the women essentially postmenopausal, with pretty good response
rates to AIs. This is a strategy that I have used clinically. If
I am going to start a premenopausal woman on an AI, I do suppress
ovarian function. Right now, you have to do that because we only
have data on AIs in postmenopausal women. This is an area that really
needs to be studied. Could you potentially treat a premenopausal
woman by increasing the dose of the AIs to get estrogen suppression?
We don’t know the answer to that.
—Stephen Jones, MD


|
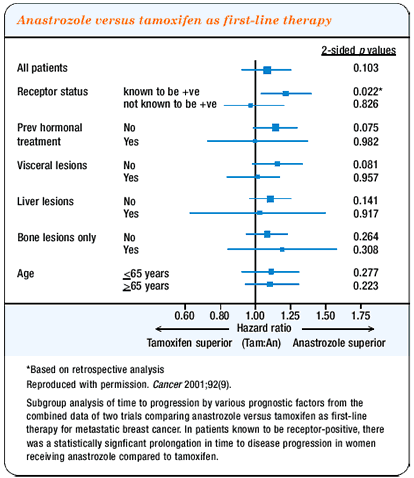
Case history continued:
The patient was continued on goserelin and switched to exemestane
51,52 but did not respond.
|
|
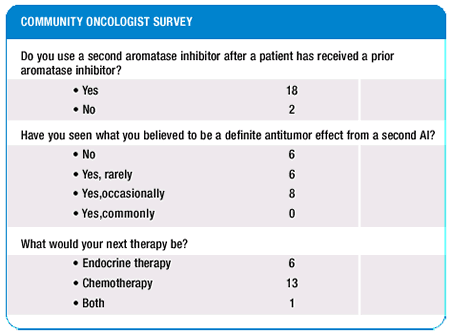
Case history continued:
After exemestane failure, the patient was placed on a dose-finding
study of paclitaxel and liposome encapsulated doxorubicin.
When she went off that study, she was treated with single
agent paclitaxel and had a partial tumor response. After having
been on paclitaxel for seven months, she developed a neuropathy.
She is currently on chemotherapy holiday.
|
CHOOSING A CHEMOTHERAPEUTIC AGENT FOR METASTATIC DISEASE
I would absolutely not have given this patient a paclitaxel-anthracycline
combination off clinical trial. Outside of a clinical trial, I sort
through this by trying to establish which single chemotherapeutic
agent was likely to produce benefit for this patient with the most
acceptable toxicity profile. Had I had the decision to make over
again in this patient and without a clinical trial available, I
probably would have given her a single-agent anthracycline.
Her neuropathy, albeit mild, has been a devastating consequence
because this patient is an accomplished seamstress. The neuropathy
has impaired her ability to do what she likes best. Unfortunately
her passion for sewing did not come to light to me until she had
already established a moderate degree of neuropathy 53,54.
Looking back, I would have selected a non-neuropathic agent such
as single-agent doxorubicin or a single-agent gemcitabine or capecitabine.
Choosing among them requires experience with the drug toxicities
and with dosing.
—Kevin Fox, MD
ALTRUISM AS A KEY COMPONENT IN TRIAL PARTICIPATION
Patients need to know that clinical trials are extraordinarily
important and the key to progress. But, they also need to know what
their choices are. Being heavy-handed about clinical trials —
as though participation is the patient’s only choice —
is not fair.
It would have been unethical for me to not tell the patient that
combination chemotherapy was a departure from my normal recommendation
under these circumstances, but this particular patient embraced
the idea very quickly. She enrolled on this clinical trial for sincerely
altruistic reasons, which was quite admirable.
Altruism is an essential ingredient in patients’ entering
clinical trials, as the benefit to an individual patient is never
as easily quantified as the benefit to all patients once the trial
is completed. Without a sense of altruism, clinical trial participation
is an uphill battle.
—Kevin Fox, MD
Page
5 of 6
Previous
page | Next page
|
